Conservation of mass in chemical reactions
December 19, 2011 at 02:03 AM | categories: uncategorized | View Comments
Conservation of mass in chemical reactions
John Kitchin
Atoms cannot be destroyed in non-nuclear chemical reactions, hence it follows that the same number of atoms entering a reactor must also leave the reactor. The atoms may leave the reactor in a different molecular configuration due to the reaction, but the total mass leaving the reactor must be the same. Here we look at a few ways to show this.
Contents
Water gas shift reaction
We consider the water gas shift reaction : % 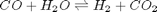 . We can illustrate the conservation of mass with the following equation:
. We can illustrate the conservation of mass with the following equation: 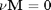 . Where
. Where  is the stoichiometric coefficient vector and
is the stoichiometric coefficient vector and 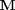 is a column vector of molecular weights. For simplicity, we use pure isotope molecular weights, and not the isotope-weighted molecular weights.
is a column vector of molecular weights. For simplicity, we use pure isotope molecular weights, and not the isotope-weighted molecular weights.
nu = [-1 -1 1 1]; M = [28; 18; 2; 44]; nu*M
ans =
0
Atomic mass balances
For any balanced chemical equation, there are the same number of each kind of atom on each side of the equation. Since the mass of each atom is unchanged with reaction, that means the mass of all the species that are reactants must equal the mass of all the species that are products! Here we look at the n C O H
reactants = [-1 -2 -2] products = [ 1 2 2] M = [12.011; 15.999; 1.0079]
reactants =
-1 -2 -2
products =
1 2 2
M =
12.0110
15.9990
1.0079
Now if we add the mass of reactants and products, it should sum to zero (since we used the negative sign for stoichiometric coefficients of reactants).
products*M + reactants*M
ans =
0
Summary
That's all there is to mass conservation with reactions. Nothing changes if there are lots of reactions, as long as each reaction is properly balanced, and none of them are nuclear reactions!
tags: reaction engineering